 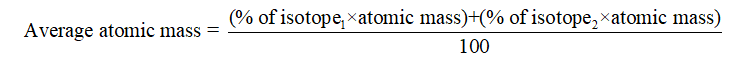
For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63, and an isotopic mass in its nuclear ground state is 62.91367 u. The isotopic mass usually differs for other isotopes and is usually within 0.1 u of the mass number. 
One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. See also: Properties of Silver Atomic Mass of Silver Silver – Properties Element Silver Atomic Number 47 Symbol Ag Element Category Transition Metal Phase at STP Solid Atomic Mass 107.8682 Density at STP 10.49 Electron Configuration 4d10 5s1 Possible Oxidation States +1 Electron Affinity 125.6 Electronegativity 1.93 1st Ionization Energy 7.5762 Year of Discovery unknown Discoverer unknown Thermal properties Melting Point 961.78 Boiling Point 2162 Thermal Conductivity 430 Specific Heat 0.235 Heat of Fusion 11.3 Heat of Vaporization 250.58 
Most silver is produced as a byproduct of copper, gold, lead, and zinc refining. The metal is found in the Earth’s crust in the pure, free elemental form (“native silver”), as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. Silver is a soft, white, lustrous transition metal, it exhibits the highest electrical conductivity, thermal conductivity, and reflectivity of any metal. Silver is a chemical element with atomic number 47 which means there are 47 protons and 47 electrons in the atomic structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
